HIV/AIDS: let’s see how far we’ve come
Lauren Fowler
Tuesday, September 1st, 2015
Now more than ever, HIV positive people are living longer and healthier lives because of access to antiretroviral therapy. Healthcare organisations are working to ensure that HIV positive people all over the world have access to the medical care they need to stay healthy. In the last few years, research into vaccine development, genetics-based approaches and novel therapies have achieved some progress and drug therapy regimens have become more effective. Public health strategies have aimed to reduce transmission, and early access to treatment has dramatically improved quality of life. With resources and funding aimed in the right directions, it will be possible to continue making significant progress towards better prevention, improved treatment options and perhaps even a cure for HIV and the elimination of the global AIDS epidemic. This article reviews some of the successes and difficulties in the scientific, research and treatment arm of combating the HIV epidemic. There is still much work to be done, but for now, let’s see how far we’ve come.

Introduction
The 2011 UNAIDS World Aids Day report concisely outlines the aims of global health efforts against HIV/AIDS: “Zero new infections. Zero discrimination. Zero AIDS-related deaths”. [1] As global citizens and future medical practitioners, it is our duty to participate in the medical issues that are of importance to the world. We should work to make the eradication of HIV/AIDS one of those key issues. This paper discusses, from the scientific perspective, some of our triumphs and tribulations with regards to combatting the complex, evasive and resourceful opponent that is the HIV virus.
The Basics
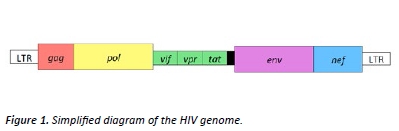
There are two main types of HIV: HIV-1 and HIV-2, with HIV-1 being the more common of the two. [2,3] HIV is a retrovirus with a high degree of variability, attributable to the error prone nature of its reverse transcriptase enzyme and its high recombination rate. [4] The HIV genome consists of a number of genes (see figure 1) including gag, pol, env and nef all of which have been used as potential antigens for the generation of vaccines. [5]
HIV primarily infects cluster-of-differentiation-4 cells (CD4) cells, including CD4 T-cells, macrophages, monocytes and dendritic cells. [6] Initially, the immune response to viral infection (including CD8 T-cell mediated killing, complement activation and antibody production) is effective at removing HIV infected cells [7], but continued immune activation and antigen presentation spreads the virus to the lymph nodes and the rest of the body. [8,9] Continued immune response to replicating virus drives development of escape mutations and ultimately overwhelms the immune system’s ability to respond. [10] As the infection progresses, the rate of destruction of infected CD4 T-cells exceeds the rate of synthesis and the CD4 count declines.
The 2008 HIV infection case definition replaces past criteria for HIV infection progression to AIDS, and divides the infection into stages reflecting the decline in immune function. [11] The stages of infection are:
Stage 1: CD4 T-cells ≥ 500 cells/mm3 (≥ 29% of total lymphocytes) with no AIDS-defining conditions.
Stage 2: CD4 T-cells 200-499 cells/mm3 (14-28% of total lymphocytes) with no AIDS-defining conditions.
Stage 3 (Progression to AIDS): CD4 T-cells <200 cells/mm3 (<14% of total lymphocytes) or the emergence of an AIDS defining condition (regardless of the CD4 T-cell count).
AIDS related conditions include a range of infections, such as esophageal Candidiasis, Cryptococcus, Kaposi sarcoma, Mycobacterium avium/ tuberculosis and Pneumocystis jirovecii pneumonia. [11] AIDS related deaths are usually due to severe opportunistic infection as the immune system is no longer able to fight basic infections. [1]
HIV is primarily transmitted via bodily fluids including blood, vaginal secretions and semen, and across the placenta, however it is not readily found in the saliva, unless there are cuts or ulcers providing access to the bloodstream. [12] Although sexual contact is the most common method of transmission, other mechanisms such as needle- stick injury, sharing needles and transfusion of HIV infected blood are far more likely to result in infection. [12] The infectivity of a person with HIV is proportional to the number of copies per mL of blood. [12]
HAART Therapy
According to UNAIDS estimates, 34 million people around the world were living with HIV in 2010, with 2/3 of the global total in Sub-Saharan Africa. [1] By the end of 2012, this had increased to 35.3 million people. [13] Now more than ever, HIV positive people are living longer and healthier lives. In part, this is due to the provision of effective treatment in the form of Highly Active Anti-Retroviral Therapy (HAART).
The first antiretroviral drug described and approved for clinical use was AZT (3’-azido-3’-deoxythymidine), a thymidine analogue. Now, there are seven categories of antiretroviral drugs, with more than 25 unique drugs. [14] HAART involves taking a combination of at least three drugs from at least two classes of antiretroviral drugs, with the aim of reconstituting lost CD4 T-cells, minimising viral load and reducing viral evolution. [15-17] The efficacy of HAART has changed HIV from a disease of significant morbidity and mortality to a manageable chronic condition. Further, HAART can minimise transmission of the virus, and prolongs the healthy lifetime of the individual by reducing viral load to undetectable levels. [18]
At present, much research is focused on investigating the timing of initiation of HAART therapy in HIV+ individuals, and the most effective combinations of drugs. [19,20] In general, those who initiate HAART therapy earlier are more likely to die at older ages and of non-AIDS causes. [21] Studies have found conflicting results when tracking the disease outcomes of patients commenced on HAART at different stages of disease. For example, a large collaborative study found that patients who are commenced on HAART with CD4 T-cell counts of 351-450 cells/mm3 have a lower rate of AIDS and death than patients whose commencement on HAART was deferred until there was further CD4 T-cell decline to below 500 cells. [18,22] Another study has shown that patients commenced on HAART at CD4 counts <500 cells/mm3 compared to deferring had slower disease progression, but this benefit was not seen with commencing HAART at CD4 T-cell counts of 500-799 cells/mm3. [19] Recent evidence has shown that early treatment enhances recovery of CD4 T-cells to normal levels. [20,23] The World Health Organisation’s HIV treatment guidelines issued in June 2013 now recommend commencing treatment when an individual’s CD4 T-cell count falls below 500 cells/mm3, or immediately on diagnosis for pregnant women, children under 5, those with HIV-associated comorbidities like tuberculosis and Hepatitis B, and for HIV- people in a serodiscordant couple with an HIV+ individual. [13]
HAART requires strict adherence, and side effects can impact on the patient’s quality of life. Long-term use is associated with toxicity, particularly to the liver, kidneys, bone marrow, brain, cardiovascular system and gastrointestinal tract. [15,24] Further, It has been noted that illnesses that typically occur with ageing appear prematurely in HIV+ patients on HAART therapy. This is thought to be only partially due to the infection, but also a side effect of the drugs involved in treatment. [25] Torres and Lewis (2014) provide an overview of what is known about premature ageing in the HIV+ patient and how this relates to HAART drugs. [25]
Non-compliance with HAART leads to the resurgence of viral replication with increased viral load and the potential for development of drug resistant mutant strains of HIV. [15,24] This can make it more difficult to treat the patient, as new drug choices may be limited and increases in viral load can lead to risk of further spread of the virus. HAART also does not purge reservoirs of latently infected cells [26], and as such, there is currently no cure for HIV.
Integration, latency and treatment challenges
There are a number of characteristics of the HIV virus that make it challenging to eradicate. HIV exhibits significant genetic diversity, both within an individual patient and within a population. As a retrovirus, HIV has an inherent ability to establish early latency within the DNA of host cells, where it remains for the lifetime of the cells and is unable to be removed by the immune system. [5]
One major development was made in 2013, when Hauber et al. reported the successful use of an antiretroviral gene therapy. A site specific recombinase (Tre) targeted to the HIV-1 long terminal repeat (LTR) was used to excise the HIV provirus from infected CD4 T-cells, functionally curing infected cells of their HIV infection, without cytopathic effects. [27] This study provides promising evidence to suggest that in future, genetically oriented antiretroviral technologies may have the potential to provide a cure for HIV infection.
Development of a prophylactic vaccine for HIV
Although it was initially believed that HIV infection was a simple illness of immunosuppression, we now know that HIV sufferers do mount strong immune responses to the HIV virus, although this response is insufficient to control the virus or eradicate the infection. [28] One of the major challenges in producing a prophylactic vaccine is the high variability of the HIV virus. There is a variation of 25-30% between subtypes of HIV-1, and 15-20% variation within any subtype. Furthermore, viral quasi-species in any infected individual can range by 10%. [29] The problem this variation causes is illustrated in natural infection, where the antibodies present in infected individuals are functional, but do not eliminate the infection due to the genetic variety seen in mutants created under the pressure of the immune system. [28,30] This variability makes it difficult to know which antigens to use to generate the required immune response to control the infection. Strategies being explored to combat this include the use of consensus sequences (fusing the most conserved portions of the virus, and trying to produce immunity to such a sequence), conserved region antigens (specifically choosing the most conserved antigens to generate immune responses) and multiple antigen cocktails (vaccination with multiple variants of one immunogen, or several different ones in combination). [31]
The first prototype HIV vaccine tested utilised purified monomeric env gp120 immunogens (a component of the virus’s surface envelope protein) in an attempt to generate virus-specific antibody responses. Unfortunately, early trials showed that this vaccine was unable to induce the production of neutralising antibodies, and did not prevent infection with HIV-1 in humans. [31,32] Since then, attempts have been made at prophylactic vaccination using a range of differing immunogens including Tat and Gag. In most cases, these vaccinations have proven safe and well tolerated, and resulted in the production of anti-HIV antibodies that may not be seen in natural infection. [33] Promising results have come from a range of trials, including the control of infected CD4 T-cells by Gag-specific CD8 T-cells, proportionally to the number of Gag epitopes recognised. [34] Success at eliciting immune responses demonstrated thus far with Gag may be to do with its relatively well-conserved sequence patterns. [34]
The STEP Study and Phambili HIV vaccine trials both used an Adenovirus 5 (Ad5) vector and gag, pol and nef immunogens, which was shown to be successful at inducing a good CD8 T-cell response. [35] Subsequent trials demonstrated that the vaccine provided no additional prevention from infection, nor a reduced early viral level. [36] Further, there was an increased rate of HIV-1 acquisition in groups of vaccinated individuals from the STEP study, most particularly in men who were already Ad5 seropositive and uncircumcised. [37]
The primary challenge in the use of viral vectors to deliver a HIV vaccine into cells is the pre-existing immunity of humans to viral vectors, leading to the neutralisation and removal of vectors from the circulation before the transfer of the immunogen to cells. In addition, vectors induce mucosal homing in T-cells, making them more susceptible to infection [38] and explaining the increased susceptibility to infection observed in the Step Study. [37,39]
At present, there is much debate over the necessary aims for a successful HIV vaccine: for example, whether to focus on the development of anti- HIV antibodies, or the induction of a CTL response. [30] Recent papers have described the ability of combinations of broadly neutralising antibodies to successfully neutralise HIV. [40,41]
MiRNAs
A new area of interest is the use of microRNAs (miRNA) as potential next generation therapeutic agents for the treatment of HIV infection and management of viremia. MiRNAs are small, noncoding RNA fragments, responsible for fine-tuning and negatively regulating gene expression. Roles for microRNAs have been found in metabolism, development and growth, and dysregulation of miRNAs have been implicated in loss of tumour suppression and development of cancer. [42,43]
The utility of miRNA-oriented technology has already been illustrated in the context of hepatitis C infection. In 2005, Jopling et al. reported that miR-122 was highly abundant in human hepatocytes, and that its presence may facilitate replication of the viral RNA, and encourage survival of the virus in the liver. [44] Since this pivotal paper, the first drug targeting a miRNA has been developed. Miraversen is a miR-122 inhibitor [45], which in human trials has been shown to exhibit dose dependent antiviral activity [46], with no dose-limiting adverse events or escape mutations observed. [47]
The first attempt to find human cellular miRNAs directly targeting the HIV genome was by Hariharan et al. (2005). [48] It was then later shown that one of the miRNAs identified was capable of inhibiting HIV nef expression, and decreasing HIV replication. [49] Further research demonstrates that cellular miRNAs potently inhibit HIV-1 production in resting primary CD4 T-cells, suggesting that cellular miRNAs are pivotal in HIV-1 latency. [50]
In 2007, Triboulet et al. reported that HIV-1 infection caused a down- regulation of specific miRNAs in mononuclear blood cells, and that this was necessary for effective viral replication. [51] Witwer et al. (2012) subsequently showed that the miRNA profiling of infected cells could be used to distinguish elite suppressors, viraemic HIV-1 patients and uninfected controls from one another [52], indicating significant changes to cellular miRNA profiles of cells when they are infected by HIV. From these studies it is evident that different cellular miRNAs modulate and are modulated by HIV infection, with different miRNAs implicated in different cells, contexts and environments. More research in this area is required, and will hopefully give rise to a new generation of therapeutic agents for HIV. The interested reader is referred to more specific reviews [53-55] for more detailed information.
Where to from here?
HIV has proven itself a formidable opponent to our aims at a global improvement in healthcare and quality of life. However, recent research gives hope that advanced treatments, better prevention and even a cure may one day be possible. It is clear that the best way to tackle HIV is by a coordinated approach, where global health strategies, clinical medicine and research work together to help eradicate this epidemic.
We have had some success in the use of novel approaches like targeting cellular miRNAs and excising HIV DNA from the human genome, and there have also been some promising results in the generation of immunity to infection through a HIV vaccine. We are constantly learning more about how HIV interacts with the host immune system, and how to overcome it. However, progress on the development of a HIV vaccine has stalled somewhat after the findings of the STEP and Philambi studies.
It is important to acknowledge the significant achievements that have been made worldwide through HIV public health campaigns. In 2012, a record 9.7 million people were receiving antiretroviral therapy, and the incidence of HIV is falling each year, with a 33% decline from 2001 to 2012. [56] However, there is still much work to be done to ensure all people are able to access HIV testing and treatment. One of the aims of the Millennium Development Goals is to provide universal access to treatment for HIV/AIDS to all those who need it, although this is yet to be achieved. [56,57]
Medicine has come a long way in the understanding and treatment of the complex and multifaceted problem that is the global AIDS epidemic. I urge medical students to be informed and interested in the HIV epidemic, and to be involved in the clinical, research and community groups tackling this multifaceted problem. With continued efforts and dedication, there is hope that in our lifetime, we may see the realisation of the ambitious aims of the 2011 UNAIDS World AIDS Day report: “Zero new infections. Zero discrimination. Zero AIDS- related deaths”.
Acknowledgements
Many thanks to my family and friends who make life fun and the effort worthwhile. This article is in acknowledgement and appreciation of the hard work and dedication of HIV/AIDS researchers everywhere.
Conflict of interest
None declared.
Correspondence
L Fowler: lauren.fowler@griffithuni.edu.au
References
[1] UNAIDS. World AIDS Day Report 2011: How to get to zero: Faster. Smarter. Better. 2011.
[2] Barre-Sinoussi F, Chermann JC, Rey F, Nugeyere MT, Chamaret S, Gruest J. Isolation of a T-lymphotropic retrovirus from a patient at risk for acquired immune deficiency syndrome (AIDS). Science. 1983; 220(4599):868-71.
[3] Clavel F, Guetard D, Brun-Vezinet F, Chamaret S, Rey M, Santos-Ferreira M, et al. Isolation of a new human retrovirus from West African patients with AIDS. Science. 1986;223(4761):343-6.
[4] Preston B, Poiesz B, Loeb L. Fidelity of HIV-1 Reverse Transcriptase. Science. 1988; 242(4882):1168-71.
[5] Chhatbar C, Mishra R, Kumar A, Singh SK. HIV vaccine: hopes and hurdles. Drug Discov Today. 2011; (21-22):948-56.
[6] Cohen M, Shaw G, McMichael A, Haynes B. Acute HIV-1 infection. New Eng J Med. 2011; 364(20):1943-54.
[7] Borrow P, Lewicki H, Hahn BH, Shaw GM, Oldstone MBA. Virus-specific CD8+ cytotoxic T-lymphocyte activity associated with control of viremia in primary human immunodeficiency virus Type 1 infection. J Virol. 1994; 68(9):6103-10.
[8] Zhang Z. Sexual transmission and propagation of SIV and HIV in resting and activated CD4+ T cells. Science. 1999; 286(5443):1353-7.
[9] Cameron P, Pope M, Granelli-Piperno A, Steinman RM. Dendritic cells and the replication of HIV-1. J Leukoc Biol. 1996; 59(2):158.
[10] Richman DD, Wrin T, Little SJ, Petropoulos CJ. Rapid evolution of the neutralizing antibody response to HIV type 1 infection. P Natl Acad Sci 2003; 100(7):4144-9.
[11] Schneider E, Whitmore S, Glynn MK, Dominguez K, Mitsch A, McKenna M. Revised surveillance case definitions for HIV infection among adults, adolescents, and children aged <18 months and for HIV infection and AIDS among children aged 18 months to <13 years. Morb Mortal Wkly Rep. 2008; 57(10):1-8.
[12] Galvin SR, Cohen MS. The role of sexually transmitted diseases in HIV transmission. Nat Rev Microbiol. 2004; 2(1):33-42.
[13] UNAIDS. GLOBAL REPORT: UNAIDS report on the global AIDS epidemic. 2013.
[14] De Clercq E. Antiretroviral drugs. Curr Opin Pharmacol. 2010 Oct;10(5):507-15.
[15] Yeni P. Update on HAART in HIV. J Hepatol. 2006; 44(1 Suppl):S100-3.
[16] Hammer SM, Saag MS, Schechter M, Montaner JSG, Schooley RT, Jacobsen DM, et Treatment for adult HIV infection: 2006 Recommendations of the International AIDS Society USA Panel. JAMA. 2006; 296(7):827-43.
[17] Lee GQ, Dong W, Mo T, Knapp DJ, Brumme CJ, Woods CK, et al. Limited evolution of inferred HIV-1 tropism while viremia is undetectable during standard HAART therapy. PloS one. 2014; 9(6):e99000.
[18] Cohen M, Chen YQ, McCauley M, Gamble T, Hosseinipour M, Kumarasamy N, et al. Prevention of HIV-1 infection with early antiretroviral therapy. New Eng J Med. 2011; 365(6):493-505.
[19] Writing Committee for the Cascade Collaboration. Timing of HAART initiation and clinical outcomes in human immunodeficiency virus type 1 seroconverters. Arch Internal Med. 2011; 171(17):1560-9.
[20] US Department of Health and Human Services. Panel on antiretroviral guidelines for adults and adolescents: guidelines for the use of antiretroviral agents in HIV-1-infected adults and adolescents. 2009; 1-161.
[21] Wada N, Jacobson LP, Cohen M, French A, Phair J, Munoz A. Cause-specific mortality among HIV-infected individuals, by CD4(+) cell count at HAART initiation, compared with HIV-uninfected individuals. AIDS. 2014; 28(2):257-65.
[22] Sterne JA, May M, Costagliola D, de Wolf F, Phillips AN, Harris R, et al. Timing of initiation of antiretroviral therapy in aids-free hiv-1-infected patients: A collaborative analysis of 18 HIV cohort studies. Lancet. 2009; 373(9672):1352-63.
[23] Le T, Wright EJ, Smith DM, He W, Catano G, Okulicz JF, et al. Enhanced CD4+ T-cell recovery with earlier HIV-1 antiretroviral therapy. New Eng J Med. 2013; 368(3):218-30. [24] Gallant J. Initial therapy of HIV infection. J Clin Virol. 2002 25:317-33.
[25] Torres RA, Lewis W. Aging and HIV/AIDS: pathogenetic role of therapeutic side effects. Laboratory invest. 2014; 94(2):120-8.
[26] Le Douce V, Janossy A, Hallay H, Ali S, Riclet R, Rohr O, et al. Achieving a cure for HIV infection: do we have reasons to be optimistic? J Antimicrob Chemoth. 2012; 67(5):1063- 74.
[27] Hauber I, Hofmann-Sieber H, Chemnitz J, Dubrau D, Chusainow J, Stucka R, et al. Highly significant antiviral activity of HIV-1 LTR-specific tre-recombinase in humanized mice. PLoS pathog. 2013 9(9):e1003587.
[28] Burton DR, Stanfield RL, Wilson IA. Antibody vs. HIV in a clash of evolutionary titans. P Natl Acad Sci USA. 2005; 102(42):14943-8.
[29] Thomson MM, Pérez-Álvarez L, Nájera R. Molecular epidemiology of HIV-1 genetic forms and its significance for vaccine development and therapy. Lancet Infect Dis. 2002; 2(8):461-71.
[30] Schiffner T, Sattentau QJ, Dorrell L. Development of prophylactic vaccines against HIV-Retrovirology. 2013; 10(72).
[31] Barouch DH, Korber B. HIV-1 vaccine development after STEP. Annu Rev Med. 2010; 61:153-67.
[32] Flynn NM, Forthal DN, Harro CD, Judson FN, Mayer KH, Para MF, rgp120 HIV Vaccine Study Group. Placebo-controlled phase 3 trial of a recombinant glycoprotein 120 vaccine to prevent HIV-1 infection. J Infect Dis. 2005; 191(5):654-65.
[33] Ensoli B, Fiorelli V, Ensoli F, Lazzarin A, Visintini R, Narciso P, et al. The preventive phase I trial with the HIV-1 Tat-based vaccine. Vaccine. 2009; 28(2):371-8.
[34] Sacha JB, Chung C, Rakasz EG, Spencer SP, Jonas AK, Bean AT, et al. Gag-specific CD8+ T lymphocytes recognize infected cells before AIDS-virus integration and viral protein expression. J Immunol. 2007; 178(5):2746-54.
[35] McElrath MJ, DeRosa SC, Moodie Z, Dubey S, Kierstead L, Janes H, et al. HIV-1 vaccine-induced immunity in the test-of-concept Step Study: a case-cohort analysis. Lancet. 2008; 372(9653):1894-905.
[36] Buchbinder SP, Mehrotra DV, Duerr A, Fitzgerald DW, Mogg R, Li D, et al. Efficacy assessment of a cell-mediated immunity HIV-1 vaccine (the Step Study): a double-blind, randomised, placebo-controlled, test-of-concept trial. Lancet. 2008; 372(9653):1881-93.
[37] Gray G, Buchbinder S, Duerr A. Overview of STEP and Phambili trial results: two phase IIb test-of-concept studies investigating the efficacy of MRK adenovirus type 5 gag/pol/nef subtype B HIV vaccine. Curr Opin HIV AIDS. 2010; 5(5):357-61.
[38] Benlahrech A, Harris J, Meiser A, Papagatsias T, Hornig J, Hayes P, et al. Adenovirus vector vaccination induces expansion of memory CD4 T cells with a mucosal homing phenotype that are readily susceptible to HIV-1. PNAS 2009; 106(47):19940-5.
[39] Robb ML. Failure of the Merck HIV vaccine: an uncertain step forward. Lancet. 2008; 372(9653):1857-8.
[40] Walker LM, Huber M, Doores KJ, Falkowska E, Pejchal R, Julien JP, et al. Broad neutralization coverage of HIV by multiple highly potent antibodies. Nature. 2011; 477(7365):466-70.
[41] Wu X, Yang ZY, Li Y, Hogerkorp CM, Schief WR, Seaman MS, et al. Rational design of envelope identifies broadly neutralizing human monoclonal antibodies to HIV-1. Science. 2010; 329(5993):856-61.
[42] Houzet L, Jeang KT. MicroRNAs and human retroviruses. Biochim Biophys Acta. 2011; 1809(11-12):686-93.
[43] Croce CM. Causes and consequences of microRNA dysregulation in cancer. Nat Rev Genet. 2009 (10):704-14.
[44] Jopling C, Yi M, Lancaster A, Lemon S, Sarnow P. Modulation of Hepatitis C virus RNA abundance by a liver-specific microRNA. Science. 2005; 309(5740):1577-81.
[45] Santaris Pharma A/S advances Miraversen, the first microRNA targeted drug to enter clinical trials, into phase 2 to treat patients infected with hepatitis C virus. Clinical Trials Week. 2010; 309.
[46] Janssen H, Reesink H, Zeuzem S, Lawitz E, Rodriguez-Torres M, Chen A, et al. A randomized, double-blind, placebo (plb) controlled safety and anti-viral proof of concept study of miravirsen (MIR), an oligonucleotide targeting miR-122, in treatment naïve patients with genotype 1 (gt1) chronic HCV infection. Hepatology. 2011; 54(1430).
[47] Janssen HL, Reesink HW, Lawitz EJ, Zeuzem S, Rodriguez-Torres M, Patel K, et al. Treatment of HCV infection by targeting microRNA. New Eng J Med. 2013; 368(18):1685-94. [48] Hariharan M, Scaria V, Pillai B, Brahmachari SK. Targets for human encoded microRNAs in HIV genes. Biochem Bioph Res Co. 2005 Dec 2;337(4):1214-8.
[49] Ahluwalia JK, Khan SZ, Soni K, Rawat P, Gupta A, Hariharan M, et al. Human cellular microRNA hsa-miR-29a interferes with viral nef protein expression and HIV-1 replication. Retrovirology. 2008; 5:117.
[50] Huang J, Wang F, Argyris E, Chen K, Liang Z, Tian H, et al. Cellular microRNAs contribute to HIV-1 latency in resting primary CD4+ T lymphocytes. Nat Med. 2007; (10):1241-7.
[51] Triboulet R, Mari B, Lin YL, Chable-Bessia C, Bennasser Y, Lebrigand K, et al. Suppression of microRNA-silencing pathway by HIV-1 during virus replication. Science. 2007; 315(5818):1579-82.
[52] Witwer KW, Watson AK, Blankson JN, Clements JE. Relationships of PBMC microRNA expression, plasma viral load, and CD4+ T-cell count in HIV-1-infected elite suppressors and viremic patients. Retrovirology. 2012; 9:5.
[53] Klase Z, Houzet L, Jeang KT. MicroRNAs and HIV-1: complex interactions. J Biol Chem. 2012; 287(49):40884-90.
[54] Fowler L, Saksena N. Micro-RNA: new players in HIV-pathogenesis, diagnosis, prognosis and antiviral therapy. AIDS Rev. 2013; 15:3-14.
[55] Gupta P, Saksena N. MiRNAs: small molecules with a big impact on HIV infection and pathogenesis. Future Virol. 2013; 8(8):769-81.
[56] UN Department of Public Information. We can end poverty: Millennium development goals and beyond 2015. Fact Sheet Goal 6: Combat HIV/AIDS, malaria and other diseases. [Internet] 2013. Available from: http://www.un.org/millenniumgoals/pdf/Goal_6_fs.pdf
[57] UN Department of Public Information. Goal 6: Combat HIV/AIDS, Malaria and Other Diseases [Internet] 2013 [cited 2014 Sept 5]. Available from: http://www.un.org/millenniumgoals/aids.shtml
