Continuous positive airway pressure for obstructive sleep apnoea improved oculogyric crises as well as psychotic symptoms in a woman with schizophrenia and developmental disability
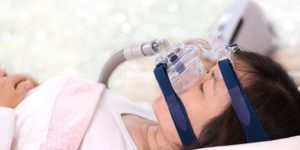
Abstract Introduction: This report highlights the risk factors and complexities of schizophrenia as well as the adverse effects of treatment. Obstructive sleep apnoea (OSA) has a notorious history of under-diagnosis …