5th Year Medicine, James Cook University
A review of the resistance mechanisms underlying dabrafenib/trametinib combined therapy in the treatment of BRAF mutant metastatic melanoma
Samantha K O’Dempsey
Monday, April 16th, 2018
5th Year Medicine, James Cook University
Abstract
Background: BRAF mutant metastatic melanoma treatment is most effective when it targets the changes induced within the mitogen-activated protein kinase (MAPK) cas-cade. However, due to the cancer’s heterogeneous nature, drug resistance predictably develops within 9-10 months, reducing treatment efficacy and producing poor patient outcomes. Understanding the mechanisms behind this acquired resistance is vital in de-vising optimal therapeutic regimens and ultimately improving the survival rate of this widespread disease.
Methods: This review examines the different resistance mechanisms that develop in BRAF mutant metastatic melanoma to prevent the durable efficacy of combined BRAF and MEK inhibitors as a treatment method. Subsequently, it evaluates possible changes that can be made to ensure therapy is made more effective in future disease management.
Results: Currently recognised resistance mechanisms include: alterations to BRAF (commonly through gene amplification), eIF4F complex mutations, changes activating the upstream regulator N-RAS or downstream effectors MEK1/2, and non-genomic al-terations. Together, these factors reactivate the MAPK cascade, despite dual MAPK in-hibition, and allow the tumour to continue to grow and metastasise unimpeded by inter-vention.
Conclusion: The ease at which contemporary treatment is being made redundant high-lights the requirement for further research into the underlying molecular aberrations, and from this, the development of new, more effective therapies into the future.
Introduction
Despite representing only 4% of all possible skin cancers, cutaneous melanoma has shown to be the most aggressive type, contributing to approximately 80% of all skin cancer related deaths (65,000 per year) [1]. It is the most common form of cancer diag-nosed in Australians aged 15-29 years, and its incidence in Caucasian populations has been increasing faster than any other malignancy over the last 30 years [2]. Five-year survival is poor for disseminated disease, from 15% to 60% in patients with either dis-tant or local metastases respectively [1,2]. Until 2011, treatment for patients with meta-static melanoma was largely ineffective, with chemotherapy having no effect on either median progression-free or overall survival [3,4]. However, an improved understanding of the underlying molecular aberrations in melanoma tumour cells led to the development of new targeted therapies, which have fortunately shown a significant clinical effect [2,5].
The genetic analysis of melanomas has revealed the clinical importance of BRAF, a pro-tein kinase that plays an essential role in activating the mitogen-actived protein kinase (MAPK)/extracellular signal related kinase (ERK)-signalling pathway [6]. Approximate-ly 40-50% of all cutaneous melanomas harbour activating BRAF mutations, although this aberration is rare in mucosal melanomas, and non-existent in the uveal form [7]. The mutation most commonly occurs in valine at codon 600, causing an increase in enzyme activity, which leads to angiogenesis, unchecked replication of cells and an ability to me-tastasise — all essential factors in tumour growth and spread [8,9]. As such, it is a vital target for drugs in reducing melanoma progression (and thus morbidity and mortality) when the diagnosis is made too late for simple excision. However, whilst treatments tar-geting a single aspect in the MAPK pathway were previously employed as a first-line defence, resistance to these therapies nearly invariably developed within 5-7 months of commencement [2,9-11].
Combined MAPK inhibition using both selective BRAF and allosteric MEK inhibitors as a method of circumventing these resistance mechanisms was subsequently consid-ered, and in 2014 met the approval of the Australian Therapeutic Goods Administration for disease treatment [3,10,12]. Initial clinical trials merging the BRAF inhibitor dabraf-enib, with MEK inhibitor trametinib, demonstrated an increased durability against drug resistance, thereby improving response rate, and enhancing progression-free and overall survival compared to single agent BRAF inhibition [7]. Following therapy initiation, 50% of patients experienced disease progression after 9-10 months, and long-term clini-cal outcomes were again impeded by the development of resistance [13,14].
Understanding of these resistance mechanisms is currently improving, however com-plete comprehension is vital if a significant reduction in melanoma mortality rates is to be achieved [5,15]. This review thus aims to provide readers with a thorough understand-ing of the resistance mechanisms that develop to the combined therapy of dabraf-enib/trametinib, and what improvements can be made to make treatment more effective in the future.
Discussion
Mechanism of action of dabrafenib/trametinib combined therapy in BRAF mu-tant metastatic melanoma
Normal MAPK function
The MAPK pathway (Figure 1) is a highly-conserved signalling cascade, essential for various cellular functions such as proliferation, differentiation and migration of cells [16-18]. Its activation is a complex process to ensure pathway regulation, and is often initiat-ed via RAS (a guanosine triposphate (GTP)-hydrolysing enzyme) binding to a GTP molecule, and leading to phosphorylation and activation of the RAF kinases [2]. These in turn phosphorylate the MEK1/2 kinases, which consequently enable ERK 1 and 2 ac-tivation [6,19,20]. ERK then proceeds to phosphorylate a plethora of cytoplasmic and nuclear substrates, which subsequently mediate the pathway’s pleiotropic effects. These include: cell cycle protein expression, proapoptotic and antiapoptotic regulation and function, and nuclear transcription factor activation [6,19-21].
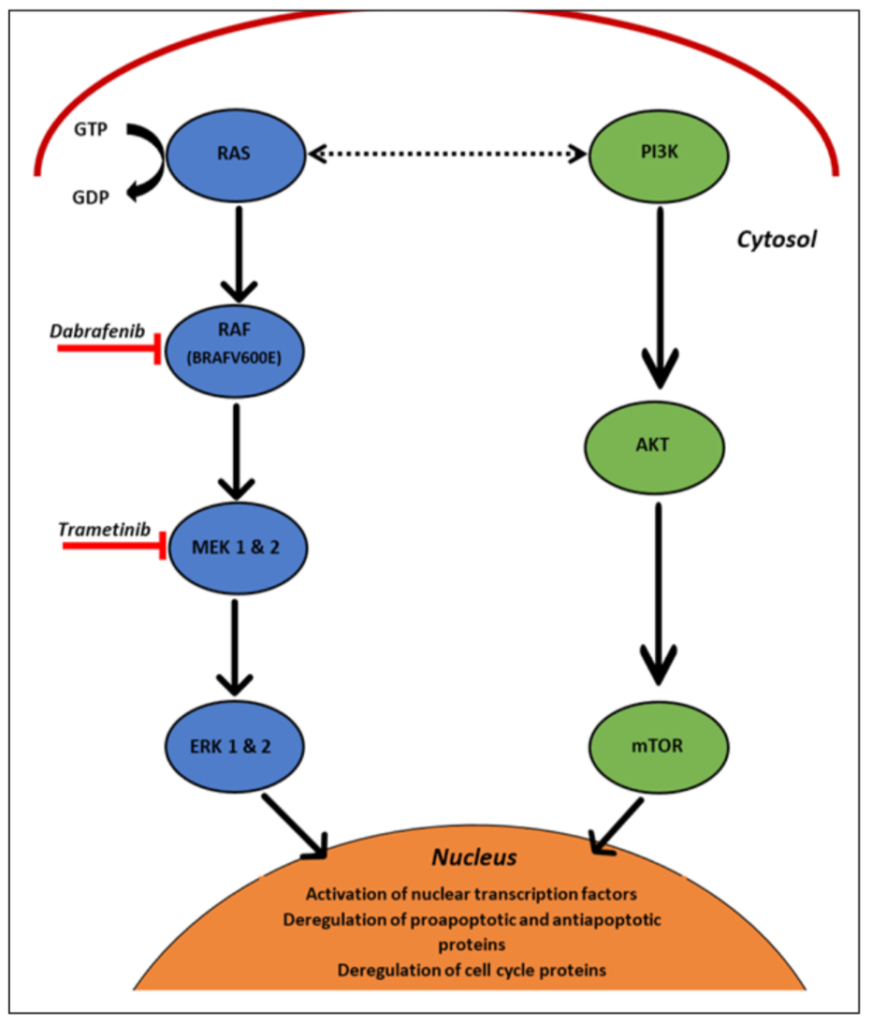
The RAF protein has three isoforms: ARAF, BRAF and CRAF [6,20,21]. As dis-cussed earlier, BRAF mutations occur at the highest frequency within most cutaneous melanomas (commonly as a V600E point mutation) and create a constitutively active BRAF molecule [23]. This subsequently leads to increased MEK and ERK function, which not only completely deregulates the cell cycle, but also increases transcription and inhibits apoptosis, thus inducing the cancer phenotype [23].
Mechanism of action of dabrafenib and trametinib
Dabrafenib is a highly potent, reversible adenosine triphosphate (ATP) competitive in-hibitor that selectively inhibits the kinase domain of mutant BRAF (Figure 1) [5,20,21]. This leads to reduced proliferation through subsequent reductions in phosphorylated ERK and thus, increased expression of apoptotic proteins and G1-phase cell cycle arrest [21]. Alternatively, trametinib is an allosteric inhibitor of MEK 1 and 2, that selectively binds and stabilises the closed, inactive conformation of the MEK enzymes, thereby re-ducing phospho-ERK concentrations and the downstream effects it has on cell prolifera-tion, growth and senescence (Figure 1) [18,24,25]. Ultimately, the combination of these two inhibitors significantly improved progression-free survival and response duration for metastatic melanoma patients when compared to monotherapy [12,14].
Mechanisms behind combined therapy resistance
The mechanisms involved in MAPK inhibitor resistance still largely require further in-vestigation. However, given the nature of combination therapy and the fact it was devel-oped to address many of the mechanisms causing acquired resistance in single target treatment, the culpable genetic aberrations are not as diverse as those seen in monothera-py [10,13,26].
There are currently several categories of mutations consistently identified as the source of MEK/ERK signalling reactivation: BRAF gene amplification, MEK1/2 mutation, and NRAS alteration [10,18,27]. There have also been a number of melanoma phenotypes recognised as possessing innate resistance, the mechanics of which are still under heavy investigation and only briefly covered within this report.
Resistance mechanism of BRAF gene amplification
BRAF copy number gains were most common, affecting 36% of all melanomas treated with combined therapy [10]. Notably, the extent of this amplification was significantly higher than seen in monotherapy resistance. Not only is this reflective of the upsurge in MAPK signalling required to overcome dual treatment, it is also indicative of increased tumour reliance on this resistance mechanism due to inhibition of other avenues [10,11,14]. Ultimately, gene amplification leads to augmented BRAF kinase concentra-tion within the cell, drastically elevating MAPK signalling by creating an excess of acti-vated MEK [8,14]. This has two vital consequences:
1. An increase in the basal level of phosho-ERK and thus increased activation of nuclear transcription factors, anti-apoptotic and cell cycle regulation proteins to cause proliferation, metastasis, and apoptosis resistance in melanoma cells [21,24].
2. An increase in the IC50 (the concentration of drug required to inhibit a biological process by half) of both trametinib and dabrafenib for inhibition of ERK phos-phorylation [6].
The latter point is explained by the mechanism of action of trametinib [14]. The drug has a significantly lower affinity for activated, phosphorylated MEK than it does for inactive MEK [10,14]. The presence of BRAF amplification and the resulting MEK hyperactiva-tion induces an excess of phosphorylated MEK, with little remaining in the drug’s fa-voured inactive conformation [10,24,25]. Overcoming this decreased affinity and ade-quately inhibiting MEK hence requires higher concentrations of trametinib [14]. Fur-thermore, because of the increased BRAF kinase concentration, therapeutic levels of dabrafenib cannot compete, and thus have an insufficient effect in inhibition [10].
Resistance mechanism of MEK1/2 mutation
Further implicated in resistance development were de novo mutations in MEK1/2, with an incidence of 26% in treatment-insensitive tumours [10,13]. The majority of MEK mu-tations employ a similar mechanism of action, with the alterations tending to occur within, or proximal to, a negative regulatory region of MEK; helix A/C sub structure [9,10,13,17]. The helix sits against the area of the kinase that binds both ATP and allos-teric inhibitors (such as trametinib). Whilst the mutations are usually located too far from the ligand to directly interact with it, they are close enough to alter the ATP binding site in a way that allosterically increases the intrinsic kinase activity of MEK [14,27]. As a result, ERK levels can be up to 20-fold higher compared to wild type MEK, increasing proliferation and apoptotic inhibition and ultimately creating an environment conducive to tumour growth [10,14]. Furthermore, over-expression of MEK causes a greater than ten-fold decrease in sensitivity to trametinib (due to mechanisms explained above), thereby increasing the concentrations required for MAPK inhibition and abrogating the effects of dabrafenib (which acts immediately upstream), and thus conferring acquired resistance to the combination therapy [10,13,27].
Coexisting NRAS mutation development
The emergence of coexisting BRAF and NRAS de novo mutations are also a possible cause for dual therapy resistance [28]. The NRAS gene encodes for the protein N-Ras, whose primary function involves regulating cell division. Its relatively high mutation rate means it contributes to the development of 15-20% of all non-uveal melanomas, and is subsequently the second most common oncogenic stimulus for cutaneous metastatic melanomas [7,28]. In around 80% of cases, these genetic aberrations involve a gain-of-function point mutation occurring at codon 61 of the NRAS gene, with the remaining alterations either affecting codons 12 or 13 [28]. This leads to a subsequent hyperactiva-tion of the RAS-RAF-MAPK and P13KT-AKT cascades, thereby increasing pro-survival protein expression, cellular proliferation, and cell cycle dysregulation [29]. The resulting synergistic effect of having two gain-of-function mutations within both NRAS and BRAF means a tumourigenic environment that supports metastasis quickly devel-ops, and the efficacy of trametinib/dabrafenib therapy is limited by the need to increase their required levels above what is therapeutically appropriate [28].
eIF4F eukaryotic translation initiation complex hyperactivation
Finally, it is important to consider downstream pathways and their influence on dual therapy drug resistance. MAPK and PI3K/AKT/mTOR signalling converge to influence the eIF4F eukaryotic translation initiation complex, a molecule nexus consisting of the eIF4G scaffolding protein, the eIF4E cap-binding protein and the eIF4A ribonucleic acid (RNA) helicase [30,31]. Its normal function involves modulating specific mitochondrial RNA translation to produce a plethora of proteins that potently regulate cell growth, pro-liferation, migration, and survival [31]. As such, excessive stimulation of the complex can alter the proteome, and ultimately give rise to the phenotypic heterogeneity of cells essential for drug resistance development, as well as tumour progression and metastasis [31,32]. There are three main mechanisms that have been identified in producing this augmented state, the first of which involves MAPK signalling reactivation through mechanisms described earlier [30]. Persistent phosphorylation of 4EBP1 (a protein that normally inhibits eIF4E binding) to permit increased translation initiation can also insti-gate hyperactivity, as can increased degradation of eIF4G through raised levels of pro-apoptotic BMF [30]. Ultimately, it is unsurprising that the enhanced formation and acti-vation of the eIF4F complex has been associated with dual therapy resistance in BRAF mutant metastatic melanoma, due to its propensity to produce the intratumoural hetero-geneity that helps enable drug resistance development [30-32].
Non-genomic resistance mechanisms
However, clinically acquired resistance to MAPK inhibition therapies cannot be fully explained through acquired genomic mechanisms, given that up to 10-20% of BRAF mutant metastatic melanoma patients never achieve a meaningful treatment response [7,27,33]. Rather, divergent transcriptional profiles exist between drug responsive cell lines and those which are intrinsically resistant, indicating that certain transcription fac-tors are innately present which can modulate melanoma response to MAPK inhibitors [7,29,33].
Differing tumour cell phenotypes and MITF
The significance of the microphthalmia-associated transcription factor (MITF) and its expression levels in treatment outcomes was one such identified transcription factor [34-36]. MITF plays a key role in the survival and differentiation of melanocytes by regulat-ing the expression of a variety of crucial melanogenic genes [35]. Whilst the majority of drug sensitive cells show high levels of MITF, both its expression and function were notably reduced within resistance lines. These cells instead tended to display elevated Nuclear Factor-κB (NF-κB) transcriptional activity, which in itself promotes melanoma progression and metastasis through pro-survival signalling [35,36]. The synergistic ef-fect of these phenotype factors creates a global transcriptional state that induces intrinsic indifference to intervention throughout all three levels of the RAF/MEK/ERK cascade. This was evidenced by the fact that, subsequent to dabrafenib/trametinib therapy, pro-gression-free survival of MITF-low/NF-κB-high melanomas was significantly shorter than the MITF-high/NF-κB-low group (median 5.0 months versus 14.5 months respec-tively) [35,36]. It should be noted however, that although this transcriptional state is cer-tainly associated with innate resistance, it can also be induced through MAPK hyperacti-vation, NF-κB induction and MITF dysregulation, thus becoming a mechanism of ac-quired resistance [36]. Ultimately, this transcriptional class distinction between BRAF mutant metastatic melanomas will aid future efforts in predicting treatment outcome and subsequently developing new therapeutic approaches for those patients unresponsive to RAF/MEK inhibition.
Further transcriptional alterations
There are a plethora of other transcriptional alterations that develop in treatment resistant tumours, frequently as the result of differential methylation of tumour cell-intrinsic cyto-sine-phosphate-guanine sites [7,28,37]. However, only the most recurrent molecular ab-errations will be discussed within this article: c-MET up expression, infra-physiologic LEF1 down expression, and YAPI signature enrichment [7,28]. Of the three, up expres-sion of c-MET not only remains the most consistently altered gene throughout treatment resistant melanomas, its degree of expression also greatly predicts patient outcomes via the mediation of MAPK-redundant survival signalling [37]. It is a receptor tyrosine ki-nase that reacts with its hepatocyte growth factor (HGF) ligand and stimulates an array of signalling pathways, ranging from proliferation to migration and invasion through activation of RAS and PI3K [37]. Evidently, its resultant hyperactivation of these path-ways ensures the level of BRAF and MEK inhibitors required to adequately control such a situation are too high to be within safe administration limits, thereby ensuring that melanoma cells carrying this mutation never respond to dual therapy [7,28].
Recurrent β-catenin-LEF1 down regulation has also shown to promote dual therapy in-sensitivity as its normally pro-apoptotic induction to MAPK inhibition is subsequently decreased [7]. Whilst this feature is essential for survival of metastatic cells, primary be-nign melanoma cells do not depend on this signalling cascade for survival. YAP1, a pro-survival factor that alters cell function through post transitional regulation, was also no-ticed to be harboured in increased quantities within MAPK inhibitor resistant tumours [7]. Given the history of known interactions between these two pathways in other bio-logical contexts, simultaneous deregulation of both β-catenin-LEF1 and YAP1 signal-ling is common, thereby resulting in an increased apoptotic threshold within melanomic cells, and thus reduced sensitivity to dual MAPK inhibition [7,28]. Given that the high-lighted transcriptional mutations are only a few of many in inducing therapy resistance, it is evident that current genomic diversity is severely limiting the long-term efficacy of dual medication.
Potential mechanisms to overcome acquired resistance in dabrafenib/trametinib combined therapy
Further MAPK pathway inhibition
MAPK-independent mechanisms of resistance were not conferred at a higher frequency in combined therapy compared to single-agent BRAF inhibitors [4,10,13]. This insinu-ates that BRAF mutant metastatic melanomas remain highly dependent on MEK/ERK signalling for tumour growth and survival, highlighting a potential avenue to increase combined treatment durability in the future and thus improve patient outcomes [10,14]. To elaborate, if other aspects of the pathway can be targeted along with BRAF and MEK inhibition, and thus the potency of MAPK inhibition further increased, it may help cir-cumvent the acquired resistance mechanisms which otherwise increase the concentration of activated MEK to levels dabrafenib/trametinib can no longer inhibit [10,12,13]. Ex-ample therapies include those targeting ERK through inhibition [4,26,27]. A preclinical study investigating this phenomenon found that BRAF/MEK/ERK inhibitor combina-tions not only delayed the emergence of acquired resistance, but they could also be used to overcome it in desensitised BRAF mutant tumours [27]. Whilst the exact reason for this is not yet clear, it is hypothesised that the ATP-competitive ERK inhibitors are less sensitive to altered conformation dynamics of activated ERK in the context of upstream oncogene amplification, and thus remain effective in inhibiting its downstream tumour-igenic effects on the cell [27]. Evidently, further refined investigation is needed into the area before more conclusive implications can be drawn, but current results allude to a hopeful future for a disease with such a poor survival rate.
Dual pathway inhibition
Furthermore, new studies have recently been released examining dual pathway inhibition [29,33]. The PI3K/AKT/mTOR pathway is an important cascade involved in signalling cellular growth, metabolism, and translation initiation (Figure 1). Along with MAPK, it is one of the most commonly altered signalling pathways in solid malignancy [33]. In melanoma cells, PI3K/AKT/mTOR has been shown to interact extensively with the MAPK pathway and potentially lead to its activation via phosphoionsitide 3-kinase (PI3K)-RAS interaction [29]. Whilst alone, it is not sufficient to completely confer re-sistance to combined therapy, PI3K has shown to contribute to earlier resistance devel-opment by modulating tumour responses to MAPK inhibitors [26,28]. Current evidence suggests that PI3K/mTOR pathway inhibition via ATP, and to a lesser extent, non-ATP competitive inhibitors can have a modest impact on both primary melanoma tumours and metastasis, diminishing the growth and proliferation of cells [29]. Therefore, the efficacy of PI3K and MAPK is being trialled in the hope it will ultimately improve patient out-comes [26,27]. Dual PI3K and MEK inhibition, the most common combination under-going investigation, currently results in only modest success and a number of relatively frequent adverse effects that include diarrhoea, nausea, pyrexia, rash, and fatigue. How-ever, this does not discount the feasibility of this dual pathway approach, but merely highlights the requirement for further investigation to improve tolerability [29]. This in-volves discerning the most optimal dosing schedule, perhaps targeting other members of the P13K/AKT/mTOR pathway, or augmenting patients with predictive factors [29].
Furthermore, the eIF4F complex (discussed earlier) sits at the junction of multiple onco-genic pathways and plays an essential role in producing the intratumoural heterogeneity that ultimately assists drug resistance development [32]. As such, its combined inhibi-tion with the MAPK pathway may provide a formidable effect in improving treatment efficacy [30]. There is currently work being done to target all three components of the complex, including: blocking eIF4E-cap interaction, interfering with eIF4E-eIF4G inter-action, inhibiting eIF4A helicase activity, and suppressing eIF4E levels — all with vary-ing degrees of success [31,32]. For example, eIF4A inhibitions (such as silvestrol) have a more potent effect compared to eIF4E in reducing global protein synthesis and thus the capability of tumour cells in developing treatment resistance, largely because cellular translation requires persistently high eIF4A concentrations, but not eIF4E, to be main-tained [30,31]. Whilst silvestrol therefore appears an advantageous agent to combine with MAPK inhibitors, it too is vulnerable to drug resistance due to overexpression of ABCB1/P-glycoprotein, thereby hampering its use in in vivo studies [30]. Ultimately, the introduction of eIF4F complex and MAPK doublet inhibitors as a possible treatment avenue is only a recent occurrence, and significantly more research is required before any definite conclusion can be established [31]. However, it theoretically can provide a potent influence in the seemingly insurmountable barrier of resistance in metastatic mel-anoma treatment, and thus provides hope for future patient outcomes [30].
Incorporation of immunomodulation in treatment
The potential of immunotherapy in treatment of metastatic melanoma has also been rec-ognised. Metastatic melanoma patients commonly display tumour-mediated immune suppression, and in the past, treatment with immunomodulatory therapies such as inter-feron alpha and high dose interleukin-2 has shown positive results [38]. With greater understanding of the immune system and its interaction with tumour cells, further inter-ventional therapies have now been developed with varying degrees of efficacy. These include monoclonal antibodies that inhibit essential immune checkpoints, such as ipili-mumab (an anti-cytotoxic T-lymphocyte-associated protein 4 inhibitor), and pembroli-zumab (an anti-programmed cell death 1 inhibitor), as well as other methods involving adoptive cell transfer [38]. Whilst it is hoped that a combination between these im-munomodulatory therapies with MAPK inhibition will improve clinical outcomes in metastatic melanoma patients, the toxic effects of such combinations currently remain unpredictable [38]. This only indicates the need for further, careful study into the dosing and timing of these dual treatments, as their potent anti-tumour activity and synergistic properties have high potential for improving patient outcomes.
Conclusion
Melanoma remains one of three cancers with an increasing mortality rate, despite exten-sive clinical investigation and the recent introduction of various novel and specific drugs into its treatment regimen [12]. This is primarily because long-term efficacy of these pharmaceuticals has been limited by the emergence of resistance in targeted cancer cells [3,5,6]. Given that the presence of BRAF mutations in metastatic melanoma has been associated with reduced survival in the absence of specific treatment, it is essential that these mechanisms are overcome to increase drug durability and thus improve patient outcomes [11]. It has been found that dabrafenib/trametinib associated resistance was primarily the result of BRAF gene amplification, MEK1/2 changes, the development of co-existing NRAS mutations, and eIF4F complex hyperactivation, as well as non-genomic alterations [10,13,14]. Together, they abrogate the effects of both drugs and cause resistance within 9-10 months of treatment commencement [21]. Possible solu-tions to overcome this include further inhibition of kinases downstream of RAF and MEK in the MAPK cascade such as ERK, as well as targeted inhibition of the heavily associated PI3K/ATK/mTOR cascade or eIF4F complex [10,26,28]. Incorporating im-munomodulatory therapies into current regimes is also a major point of consideration [36]. Ultimately, full comprehension of the factors influencing combination therapy re-sistance is fundamental, for only with understanding can solutions be developed, and the currently pitiable patient outcomes for BRAF mutant metastatic melanoma be improved.
As medical students enter the workforce, combination therapies will likely be the fore-front of metastatic melanoma treatment and only the beginning of the trend towards mu-tation-specific cancer management. As such, is it essential to have not only a strong un-derstanding of the basic pathways affected by these cancers, but the clinical relevance the short-term efficacy of these drugs have for patients. This need to be informed is only further augmented by the increasingly high incidence of melanoma in Australia.
Limitations of review
The major limitation of this literature review relates to the contemporary nature of the topic. With primary journal and review articles assessing the efficacy of dabrafenib/ tra-metinib still being released, investigations into the resistance mechanisms behind these results, and methods to overcome them, have only recently commenced. Consequently, there were a restricted number of articles available to examine which may affect the va-lidity of conclusions within this review when further information is made available. Fur-thermore, the sample sizes within the available primary journal articles were small [10]. Whilst the overlap in implicated mutations and possible solutions suggests valid results, it is possible that further vital mechanisms were missed because of the small sample size. Finally, it should be noted that whilst this review only focuses on dabrafenib/trametinib dual therapy, in actuality there are three BRAF/MEK doublets that have shown clinical benefit (dabrafenib/trametinib, vemurafenib/cobimetinib, encorafenib/binimetinib) [39].
Conflict of interest
None declared.
References
[1] Jemal A, Ma J, Siegel R, Zou Z. Cancer statistics, 2014. CA Cancer J Clin. 2014;64(1):9-29.
[2] Jang S, Atkins MB. Which drug, and when, for patients with BRAF-mutant melanoma? Lancet Oncol. 2013;14(2):60-9.
[3] Spagnolo F, Ghiorzo P, Orgiano L, Pastorino L, Picasso V, Tornari E, et al. BRAF-mutant melanoma: treatment approaches, resistance mechanisms, and diagnostic strategies. Onco Targets Ther. 2015;8:157-68.
[4] Middleton MR, Grob JJ, Aaronson N, Fierlbeck G, Tilgen W, Seiter S, et al. Random-ised phase III study of temozolomide versus decarbazine in the treatment of patients with advanced metastatic malignant melanoma. J Clin Oncol. 2000;18(1):2351-2.
[5] Sullivan RJ, Flaherty KT. New strategies in melanoma: entering the era of combinatorial therapy. Clin Cancer Res. 2015;21(11):2424-35.
[6] Wangari-Talbot J, Chen S. Genetics of melanoma. Front Genet. 2012;3:330.
[7]. Hugo W, Shi H, Sun L, Piva M, Song C, Kong X, et al. Non-genomic and immune evolution of melanoma acquiring MAPKi resistance. Cell. 2015;162(6):1271-85.
[8] Ascierto PA, Kirkwood JM, Grob JJ, Simeone E, Grimaldi AM, Maio M, et al. The role of BRAF V600 mutation in melanoma. J Transl Med. 2012;10:85.
[9] Maurer G, Tarkowski B, Baccarini M. Raf kinases in cancer-roles and therapeutic op-portunities. Oncogene. 2011;30(32):3477-88.
[10] Long GV, Fung C, Menzies AM, Pupo GM, Carlino MS, Hyman J, et al. Increased MAPK reactivation in early resistance to dabrafenib/trametinib combination therapy of BRAF-mutant metastatic melanoma. Nat Commun. 2014;5:5694.
[11] Long GV, Menzies AM, Nagrial AM, Haydu LE, Hamilton AL, Mann GJ, et al. Prognostic and clinicopathologic associations of oncogenic BRAF in metastatic melanoma. J Clin Oncol. 2011;29(10):1239-46.
[12] Long GV, Stroyakovskiy D, Gogas H, Levchenko E, de Braud F, Larkin J, et al. Dabrafenib and trametinib versus dabrafenib and placebo for Val600 BRAF-mutant mela-noma: a multicentre, double-blind, phase 3 randomised controlled trial. Lancet. 2015;386(9992):444-51.
[13] Wagle N, Van Allen EM, Wargo JA, Garraway LA. MAP kinase pathway alterations in BRAF-mutant melanoma patients with acquired resistance to combined RAF/MEK inhi-bition. Cancer Res. 2014;4:61.
[14] Villanueva J, Infante JR, Krepler C, Reyes-Uribe P, Samanta M, Chen H, et al. Con-current MEK2 mutation and BRAF amplification confer resistance to BRAF and MEK in-hibitors in melanoma. Cell rep. 2013;4(6):1090-9.
[15] Flaherty KT, Infante JR, Daud A, Gonzalez R, Kefford RF, Sosman J, et al. Combined BRAF and MEK inhibition in melanoma with BRAF V600 mutations. N Engl J Med. 2012;367(18):1694-703.
[16] Flaherty KT, Puzanov I, Kim KB, Ribas A, McArthur GA, Sosman JA, et al. Inhibi-tion of mutated, activated BRAF in metastatic melanoma. N Engl J Med. 2010;363(9):809-19.
[17] Ahronian LG, Sennott EM, Van Allen EM, Wagle N, Kwak EL, Faris JE, et al. Clini-cal acquired resistance to RAF inhibitor combinations in BRAF-mutant colorectal cancers through MAPK pathway alterations. Cancer Discov. 2015;5:358.
[18] McCubrey JA, Steelman LS, Chappell WH, Abrams SL, Wong EW, Chang F, et al. Roles of the RAF/MEK/ERK pathway in cell growth, malignant transformation and drug resistance. Biochim Biophys Acta. 2007;1773(8):1263-84.
[19] Solit DB, Rosen N. Towards a unified model of RAF inhibitor resistance. Cancer Dis-cov. 2014;4(27):27-30.
[20] Santarpia L, Lippman SL, El-Naggar AK. Targeting the mitogen-activated protein ki-nase RAS-RAF signaling pathway in cancer therapy. Expert Opin Ther Targets. 2012;16(1):103-19.
[21] Wang AX, Qi XY. Targeting RAS/RAF/MEK/ERK signalling in metastatic melano-ma. Mol Cell Biol. 2013;65(9):748-58.
[22] Read J. Recent advances in cutaneous melanoma: towards a molecular model and tar-geted treatment. Australas J Dermatol. 2013;54(3):163-72.
[23] Byron SA., Loch DC, Wellens CL, Wortmann A, Wu J, Wang J, et al. Sensitivity to the MEK inhibitor E6201 in melanoma cells is associated with mutant BRAF and wild type PTEN status. Mol Cancer. 2012;11(75):576-7.
[24] Corcoran RB, Dias-Santagata D, Bergethon K, Settleman J, Engelman J, Iafrate JA. BRAF gene amplification can promote acquired resistance to MEK inhibitors in cancer cells harbouring the BRAF V600E mutation. Sci Signal. 2010;3(149):84.
[25] Duncan JS, Whittle MC, Nakamura K, Abell AN, Midland AA, Zawistowski JS, et al. Dynamic reprogramming of the kinome in response to targeted MEK inhibition in triple negative breast cancer. Cell. 2012;149(2):307-21.
[26] Carlino MS, Todd JR, Gowrishankar K, Mijatov B, Pupo GM, Fung C, et al. Differ-ential activity of MEK and ERK inhibitors in BRAF inhibitor resistant melanoma. Mol On-col. 2014;8(3):554.
[27] Emery C, Vijayendran K, Zipser M, Sawyer A, Niu L, Kim J. MEK 1 mutations con-fer resistance to MEK and B-RAF inhibition. Proc Natl Acad Sci USA. 2009;106(48):20411-6. doi: 10.1073/pnas.0905833106.
[28] Moriceau G, Hugo W, Hong A, Shi H, Kong X, Yu C, et al. Tunable-combinatorial mechanisms of acquired resistance limit the efficacy of BRAF/MEK co-targeting but result in melanoma drug addiction. Cancer Cell. 2015;27(2):240-56. doi:10.1016/j.ccell.2014.11.018
[29] Shimizu T, Tolcher A, Papadopoulos K, Beeram M, Rasco D, Smith L. The clinical effect of the dual-targeting strategy involving PI2K/AKT/mTOR and RAS/MEK/ERK pathways in patients with advanced cancer. Clin Cancer Res. 2012;18(8);2316–25. doi:10.1158/1078-0432.CCR-11-2381
[30] Boussemart L, Malka-Mahieu H, Girault I, Allard D, Hemmingsson O, Tomasic G, et al. eIF4F is a nexus of resistance to anti-BRAF and anti-MEK cancer therapies. Nature. 2014; 513(7516):105-9. doi:10.1038/nature13572
[31] Pelletier J, Graff J, Ruggero D, Sonenberg N. Targeting the eIF4F translation initiation complex: a critical nexus for cancer development. Cancer Res. 2015;75(2):250-63. doi:10.1158/0008-5472.CAN-14-2789
[32] Kemper K, de Goeje P, Peeper D, van Amerongen R. Phenotype switching: tumour cell plasticity as a resistance mechanism and target for therapy. Cancer Res. 2014;74(21):5937-41. doi:10.1158/0008-5472.CAN-14-1174
[33] Hatzivassiliou G, Liu B, O’Brien C, Spoerke JM, Hoeflich KP, Haverty PM, et al. ERK inhibition overcomes acquired resistance to MEK inhibitors. Mol Cancer Ther. 2012;11(5):1143-54. doi:10.1158/1535-7163.MCT-11-1010
[34] Kinkade CW, Castillo-Martin M, Kuter AP, Yan J, Foster TH, Gao H, et al. Targeting AKT/mTOR and ERK MAPK signalling inhibits hormone-refractory prostate cancer in a preclinical mouse model. J Clin Invest. 2008;118(9):3051-64. doi:10.1172/JCI34764
[35] Muller J, Krijgsman O, Tsoi J, Robert L, Hugo W, Song C. Low MITF/AXL ratio predicts resistance to multiple targeted drugs in melanoma. Nat Commun. 2014;5:5712. doi:10.1038/ncomms6712
[36] Konieczkowski D, Johannessen C, Abudayyeh O, Kim J, Cooper Z, Piris A. A mela-noma cell state distinction influences sensitivity to MAPK pathway inhibitors. Cancer Dis-cov. 2014;4(7):816-27. doi:10.1158/2159-8290.CD-13-0424
[37] Organ S, Tsao M, de Bono J. An overview of the c-MET signalling pathway. Ther Adv Med Oncol. 2011;3(1):S7-19. doi:10.1177/1758834011422556
[38] Kirkwood J, Tarhini A, Panelli M, Moschos S, Zarour H, Butterfield L. Next genera-tion of immunotherapy for melanoma. J Clin Oncol. 2008; 26(20):3445-55. doi:10.1200/JCO.2007.14.6423
[39] Sullivan R, LoRusso P, Boerner S, Dummer R. Achievements and challenges of mo-lecular targeted therapy in melanoma. Am Soc Clin Oncol Educ Book. 2015:177-86. doi: 10.14694/EdBook_AM.2015.35.177
